HIMS stock is drawing renewed attention after a regulatory signal around peptide therapy introduced a potential expansion path for the company. According to StockChaser, the FDA's move does not indicate approval - but it does create a framework that could reshape how peptides are accessed and produced by compounders. That distinction matters, and the market is responding to direction, not certainty.
This is not an approval event. It reflects a shift toward clearer rules that could allow compounders to produce peptides within a defined framework - and that changes the calculus for companies already positioned in this space.
HIMS Stock and the FDA Signal: What It Does and Does Not Mean
The regulatory development does not confirm that peptide therapies are now approved for broader use. What it does is define a possible pathway - one where compounders could operate under clearer guidelines if the regulatory environment continues moving in this direction. For investors watching HIMS Stock Oversold With Support at $15 and $13.60, the setup is familiar: a catalyst based on potential rather than confirmed outcome.
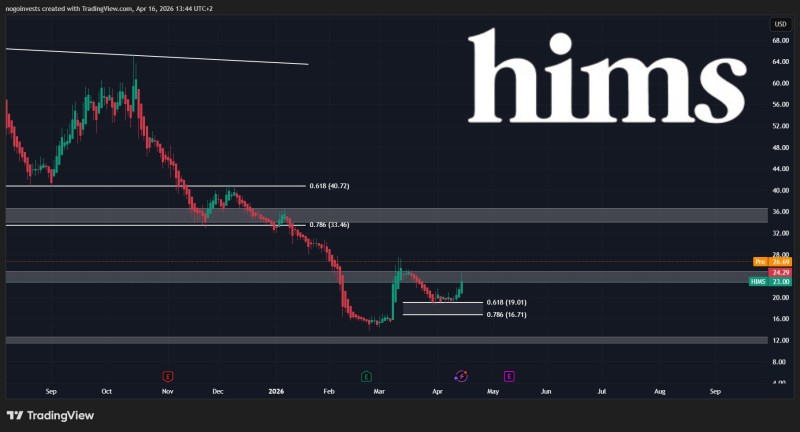
That framing is important. Reacting to direction rather than certainty is exactly what this moment calls for - and that dynamic is already visible in how the stock has been trading.
HIMS Already Has Peptide Manufacturing Capacity in Place
One of the more concrete elements here is that Hims & Hers has already committed capital to peptide manufacturing facilities. Rather than waiting for regulatory clarity to arrive and then scrambling to build infrastructure, the company made those investments in advance. If the pathway becomes actionable, HIMS enters it with production capacity already running - a structural advantage that takes time and money to replicate.
The company has already built capacity tied to this category. That is not a reaction - it is positioning. And positioning ahead of regulatory change is exactly the kind of move that compounds over time if the thesis plays out.
Investors tracking the broader volatility in the name - including the HIMS Stock Down 18% as Institutional Holdings Hit 240M Shares episode - will recognize that the company has been building through drawdowns rather than pulling back.
5 HIMS Peptide Categories That Could Expand With Regulatory Clarity
The categories in question span a wide range of therapeutic applications. If access is formalized, the addressable market for each could grow significantly:
- Weight-related compounds such as MOTS-c
- Muscle-related peptides including BPC-157 and TB-500
- Healing-focused compounds like BPC-157 and KPV
- Brain-related peptides such as Semax
- Longevity-focused compounds like Epitalon
Taken together, these categories illustrate just how broad the potential expansion could be - provided the regulatory framework continues to develop in a direction that allows formal access.
Peptides represent a high-margin category that could give HIMS another lever to monetize its existing customer base without needing to acquire entirely new audiences. The infrastructure question is already answered - the regulatory one is not.
HIMS Stock Total Addressable Market Could Grow if Pathway Becomes Actionable
The key implication for HIMS stock is straightforward: peptides are a high-margin category, and if regulatory clarity progresses, the company gains access to a market it is already operationally ready to serve. That is a meaningful expansion of total addressable market built on top of an existing customer base - without the cost of building that base from scratch.
At the same time, the setup remains conditional. Execution depends entirely on how the regulatory picture evolves. For context on where the stock has been and what technical levels matter now, see HIMS Stock Drops to $27 Before Showing Bullish Divergence Signal.
 Saad Ullah
Saad Ullah

 Saad Ullah
Saad Ullah


